Ontario Animal Health Network (OAHN) Bovine Expert Network Quarterly Veterinary Report
Surveillance Updates
HPAI in Idaho, USA
After almost four months with no detections, highly pathogenic avian influenza (HPAI H5N1) was confirmed in five Idaho dairy herds in April 2026. The detections prompted quarantines, and investigation suggests local circulation rather than a new spillover event.
Epizootic hemorrhagic disease (EHD) in Oregon, USA
Epizootic hemorrhagic disease (EHD) has been confirmed as the cause of severe neurological abnormalities in calves from multiple cattle herds in Wallowa County, Oregon. The vector‑borne virus, transmitted by biting midges and more commonly affecting wild deer, has resulted in stillbirths and calves born with extensive brain necrosis, with an estimated 40–50 cases reported locally. There is no available vaccine or treatment, and control efforts are focused on reducing insect exposure in affected areas.
Bovine tuberculosis in Michigan, USA
In February 2026, Michigan confirmed bovine tuberculosis in a dairy herd in Charlevoix County after an infected adult cow was identified at a processing plant and traced back to its herd of origin. Subsequent whole‑herd testing identified additional TB‑positive animals, prompting quarantines and a formal epidemiologic investigation. The detection occurred outside Michigan’s Modified Accredited Zone, emphasizing ongoing exposure risk from infected wildlife, particularly free‑ranging white‑tailed deer.
Global Expansion of Foot and Mouth Disease Serotype SAT 1
Over the past year, foot‑and‑mouth disease virus (FMDV) serotype SAT‑1 has shown unprecedented geographic expansion, moving beyond its historic confinement to sub‑Saharan Africa. Since 2025, SAT‑1 has been detected across parts of Western Asia, North Africa, and southeastern Europe, with two cocirculating topotypes identified. Topotypes are genetically and geographically distinct groups within a virus serotype, defined by characteristic patterns in viral genome sequences that reflect regional evolutionary lineages. Because immunity and vaccines are serotype‑specific, the spread of SAT‑1 into largely naïve livestock populations has significantly heightened transboundary disease risk and surveillance concern.
Q4 Bovine Data from the Animal Health Laboratory
Stats for Q4
There were a total of 1551 bovine submissions in Q4 2025-6. Of these, a
total of 187 submissions to the AHL had a pathology component:
- 63 postmortem cases and 124 send-in cases (including 33 meat inspection)
- 71 dairy, 58 beef, 1 Watusi, 25 not-specified
- Animals with the commodity not specified were up significantly from 7 in Q3.
- 128 submissions had a definitive or presumptive diagnosis, 27 did not have a specific diagnosis (8 abortion work-ups, 4 young calves, 10 older calves, 5 adults)
- The majority of these cases were send-in. For most submissions, a limited set of tissues had been sampled for histopathology or for culture, and history was often minimal.
- Insufficient clinical history (none) was associated with 5 submissions.
| Young calves (under 2
months) |
37 |
| Older calves (2 months-2
years) |
62 |
| Adult cattle (> 2 years)
**one case counted twice-fetus and adult cow submitted together |
34 |
| Reproductive Loss | 22 |
| Meat inspection | 33 |
Pathology Cases of Interest
Salmonella Dublin Abortion in Dairy Heifers
History: 90 pregnant heifers kept in barn with outside lot on farm separate from main dairy. Outside lot has a low area with some standing water in recent wet weather. Several heifers aborted in the previous week. The remainder of heifers appear healthy and in good condition.
Pathology: Limited gross and histologic lesions except for mild epicarditis in the fetus.
Ancillary testing: Bacterial culture grew a moderate to abundant amount of Salmonella Dublin from fetal lung, abomasal content, and placenta. PCR was negative for BHV-1, BVDV, Leptospira spp, and Neospora.
Summary: Despite the limited histologic lesions in this case, the abundant S. Dublin cultured from both the placenta and fetal tissues was considered significant in this case. Maternal bacteremia with spread to the fetus was considered most likely. Follow-up testing from this farm detected S. Dublin in a tissue sample and several blood samples of adult cows.
Iron Deficiency Anemia in Milk-Fed Calves
History: History of calves that were previously drinking 6-9L/day of whole milk via the bottle in individual housing. When moved to group housing on a robot feeder (whole milk), intakes dropped to below 6L/day, calves were sluggish and not growing optimally, were not aggressive at the feeder and aborted visits increased. Previously treated with metacam. One calf was sacrificed, post mortem showed white lungs, slightly pale muscles and organs, potentially slightly enlarged heart size, micro-abscesses of both kidneys.
Pathology: Severe suppurative nephritis noted histologically, no further testing pursued related to this. No histological correlation for low iron or anemia.
Ancillary testing: Trace mineral testing did confirm low iron levels.
Summary: Histologic correlates for iron deficiency and anemia can be limited. Trace mineral testing was essential for this case. Feeding whole milk in calves is linked to this condition, but unusual as a pathology submission.
Proteus mirabilis-Associated Cystitis and Sepsis in a Feedlot Steer
History: Feedlot cattle experiencing unexplained sudden deaths, with six animals found dead over the past two months. The primary concerns include clostridial enterotoxaemia, bloat and AIP. Two steers found dead this week, one submitted.
Pathology: This animal has severe cystitis and nephritis with peritonitis grossly. Histologically there was marked neutrophilic and fibrinous inflammation of the bladder, ureters, and kidneys with intralesional Gram negative bacteria.
Ancillary testing: Bacterial culture grew Proteus mirabilis from the lung and the urinary bladder.
Summary: This animal had severe urinary tract disease due to Proteus mirabilis, and culture of this bacteria from the lung as well as histologic lesions of sepsis support systemic spread of this opportunistic bacterial agent. Proteus mirabilis is a relatively common opportunistic pathogen of the urinary tract of cattle and can be associated with other presentations including diarrhea in calves and mastitis, pneumonia, or endometritis in adults. Severe disease is typically associated with stress/immunosuppression.
Salmonella Report
In total, 224 bovine submissions had bacterial culture performed (non-milk), generating 316 cultures. Salmonella spp. were isolated from 8 submissions, representing an estimated 7 premises. There were 16 bovine submissions that had Salmonella Dublin PCR performed, generating 1 positive submission, also captured in the culture data.
Salmonella Dublin was isolated from 5 submissions, representing an estimated 4 premises, all believed to be new premises (see table). Other serovars including S. Arizonae, S. Agona and S. Typhimurium were limited to single fecal submissions.
| Age Group/Commodity | S. Dublin Clinical Presentation | Test method |
| Fetus | Abortion | Culture |
| 2 weeks –Dairy | High morbidity/mortality (~60%) | Culture |
| 1 month – Dairy | Septicemia | Culture + PCR |
| 6 weeks – Dairy | Sick, scours | Culture |
| Adult (2 yr) – Dairy | Follow-up from known exposure | Culture |
Bovine Viral Diarrhea Virus
In total, 24 submissions generated 91 samples for BVDV PCR testing. This yielded no positive samples. BVDV immunohistochemistry was performed on one pathology submission; BVDV was not detected.
This summary has been compiled by Dr. Dominique Comeau, Animal Health Laboratory (AHL) from diagnostic submissions to the AHL Guelph and Kemptville locations.
OAHN DATA Summary: Dairy Respiratory Testing Results 2021-2025
This quarter, bovine respiratory disease submissions from dairy cattle were analyzed and the positive diagnostic results are presented. Only submissions that were identified as dairy are included, bovine with no commodity reported and dairy with no age have been excluded.
Dairy: Number of Detections on Lung Culture for BRD Pathogens of Interest
145 samples from dairy cattle < 2 months, 148 samples from dairy cattle ≥ 2 months and l< 2 years, and 99 samples from dairy cattle ≥ 2 years were submitted for lung culture. BRD pathogens of interest are presented below. Additional bacterial species were identified on an isolated basis; however, these organisms are not considered primary respiratory pathogens and were not considered of clinical concern in this context.
| Bacteria | Number of Detections
< 2 months |
Number of Detections
≥ 2 months – 2 years |
Number of Detections
≥ 2 years |
| Bibersteinia trehalosi | 2 | 3 | 4 |
| Histophilus somni | 3 | 10 | 3 |
| Mannheimia haemolytica | 4 | 11 | 3 |
| Mannheimia haemolytica Type 1 | 2 | 2 | 1 |
| Mannheimia haemolytica Type 2 | 5 | 22 | 12 |
| Mycoplasma bovis | 1 | 2 | 6 |
| Pasteurella multocida | 21 | 37 | 18 |
| Salmonella Gp D (Salmonella Dublin) | 18 | 17 | 1 |
| Trueperella pyogenes | 26 | 30 | 37 |
Dairy: Number of Detections on PCR/ IHC for BRD Pathogens of Interest
| < 2 months
% Positivity (n pos/n tested) |
≥ 2 months – 2 years
% Positivity (n pos/n tested) |
≥ 2 years
% Positivity (n pos/n tested) |
|
| Bovine herpesvirus 1 PCR | 6.42% (7/109) | 0.72% (1/138) | 1.62% (2/123) |
| Bovine parainfluenzavirus 3 PCR | 5.81% (5/86) | 1.56% (2/138) | 0% (0/93) |
| Bovine respiratory syncytial virus PCR | 6.98% (6/86) | 17.19% (22/128) | 10.75% (10/93) |
| Bovine coronavirus PCR | 42.50% (17/40) | 21.31% (13/61) | 0% (0/51) |
OAHN DATA Summary: Beef Respiratory Testing Results 2021-2025
This quarter, bovine respiratory disease submissions from cattle identified as “Meat” in submissions were analyzed and the positive diagnostic results are presented. Bovine submission with no commodity reported and meat with no age recorded have been excluded.
Beef Number of Detections on Lung Culture for BRD Pathogens of Interest
183 samples from beef cattle < 6 months and 93 samples from beef cattle ≥ 6 months were submitted for lung culture. BRD pathogens of interest are presented below. Several additional bacterial species were identified on an isolated basis; however, these organisms are not considered primary respiratory pathogens and were considered not of clinical concern in this context.
| Bacteria | Number of Detections
< 6 months |
Number of Detections
≥ 6 months |
| Bibersteinia trehalosi | 5 | 3 |
| Histophilus somni | 34 | 13 |
| Mannheimia haemolytica | 9 | 5 |
| Mannheimia haemolytica Type 1 | 2 | 1 |
| Mannheimia haemolytica Type 2 | 12 | 8 |
| Mycoplasma bovis | 8 | 5 |
| Pasteurella multocida | 30 | 14 |
| Salmonella Gp D (Salmonella Dublin) | 17 | 1 |
| Trueperella pyogenes | 27 | 13 |
Beef Number of Detections on PCR/ IHC for BRD Pathogens of Interest
| < 6 months
% Positivity (n pos/n tested) |
≥ 6 months
% Positivity (n pos/n tested) |
|
| Bovine herpesvirus 1 PCR | 3.57% (6/168) | 5.04% (6/119) |
| Bovine parainfluenzavirus 3 PCR | 1.40% (2/143) | 1.90% (2/105) |
| Bovine respiratory syncytial virus PCR | 17.48% (25/143) | 11.43% (12/105) |
| Bovine coronavirus PCR | 23.53% (16/68) | 18.52% (10/59) |
Mastitis Pathogens from Milk Cultures Submitted to the Animal Health Laboratory in 2025
Summary provided by Dr. Tanya Rossi and Dr. Durda Slavic
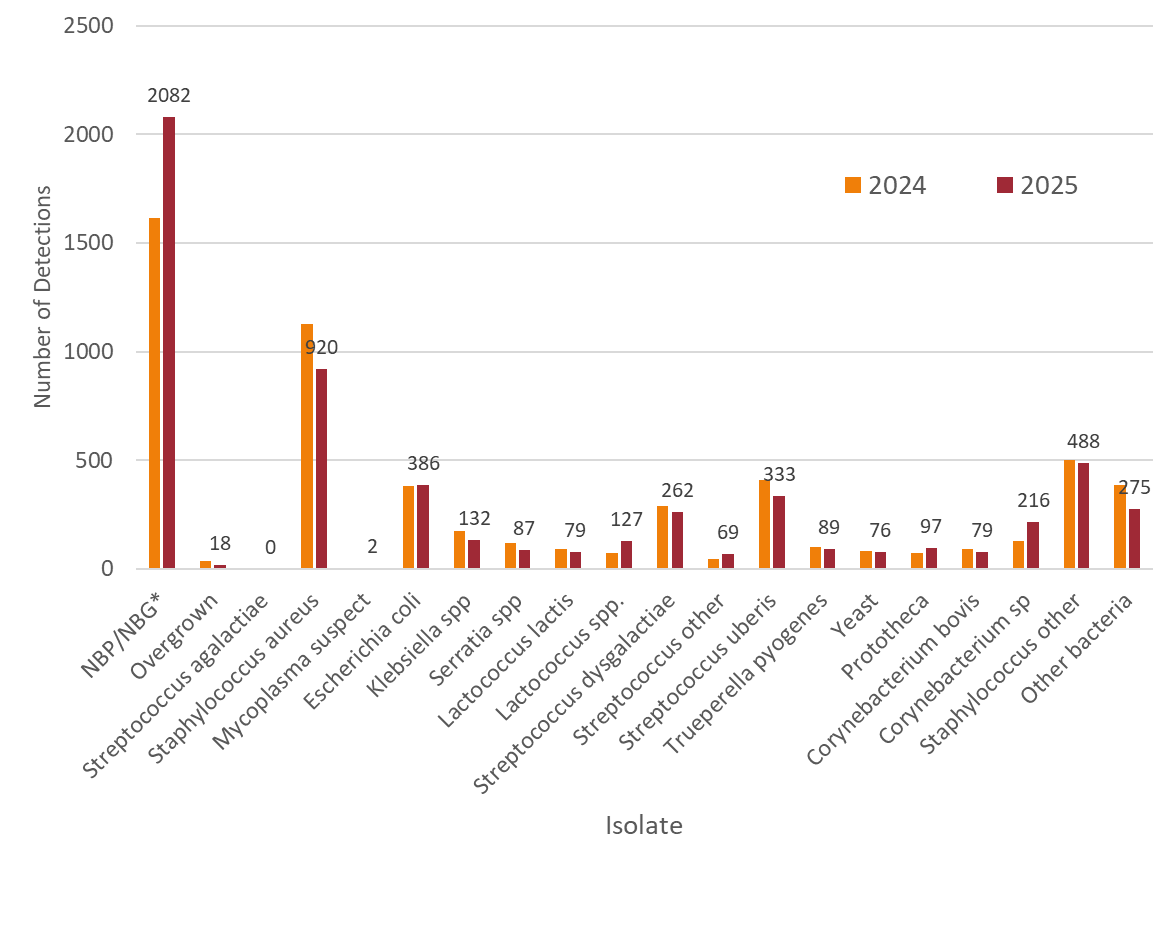
In 2025, there were 5,154 individual milk samples sent to the AHL from 1,560 submissions compared to 4,983 individual milk samples from 1,699 submissions in 2024. Data labels from the 2025 milk culture detections are included in the figure to aid with interpretation. The bar graphs with the number of detections in 2024 are included for reference.
Salmonella Dublin 2025
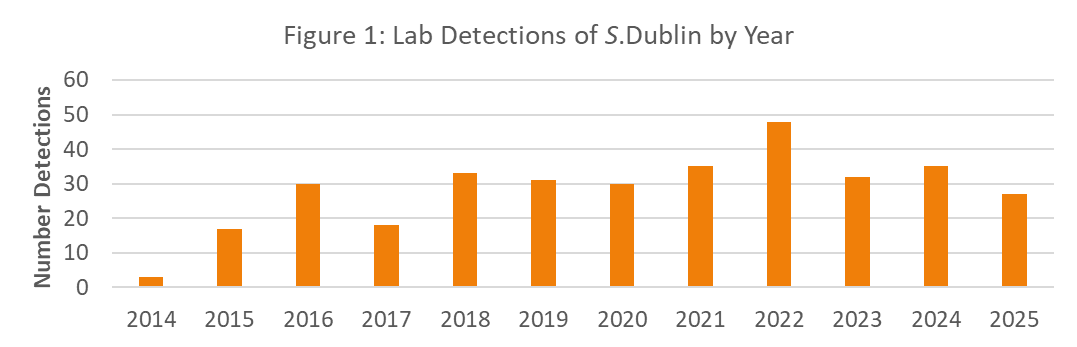
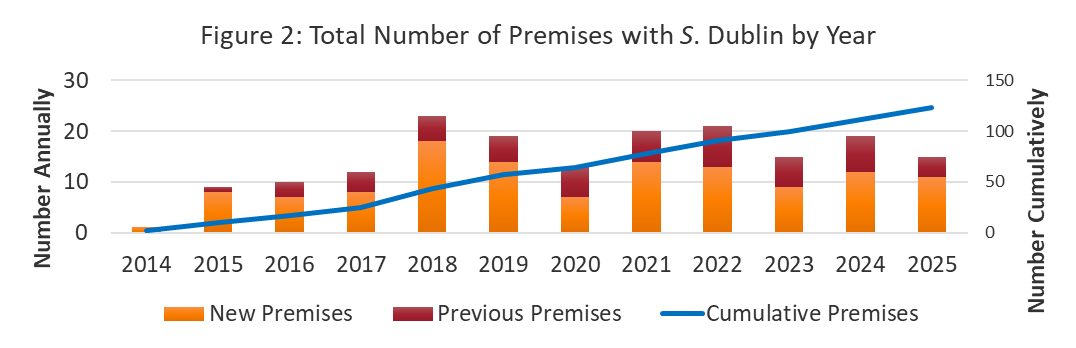
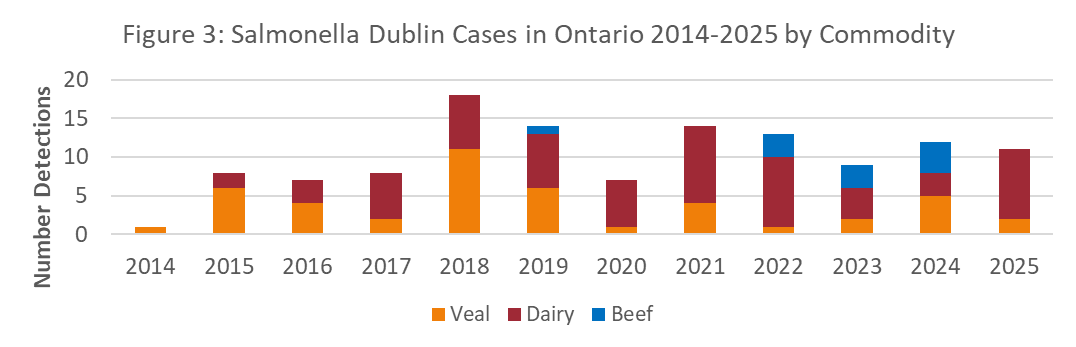
During 2025, there were 27 lab diagnostic submissions to the AHL where Salmonella Dublin was detected on bacterial culture or PCR test (Figure 1). The submissions are estimated to represent 15 unique farms and for 11 farms, this was their first diagnosis of S. Dublin (Figure 2). As of December 31, 2025, there are estimated to be 123 unique premises in the laboratory dataset that have had S. Dublin. The new farm detections in 2025 were predominantly from farms labelled as dairy (Figure 3).
Salmonella Dublin – What’s New in Research in 2025
Calf Movement Networks as Drivers of Salmonella Dublin Risk
Livestock dealer and calf assembly facilities represent critical intersections for Salmonella Dublin introduction and spread. Environmental surveillance in a Midwestern U.S. commingling facility demonstrated intermittent but repeated S. Dublin detection, most strongly associated with instability in calf sourcing (frequent change in source farms or regions) rather than volume of calves moved. This project demonstrated short‑term introduction events rather than constant contamination risk can maintain transmission risk and the absence of a detection at a single time point does not indicate absence of risk.
Early Transmission in Calf‑Raising Facilities
Cohort studies of calves entering commercial calf‑raising systems demonstrate early exposure and within‑facility transmission of S. Dublin, with associated impacts on growth and increased respiratory and enteric disease. These facilities function as amplification points even without obvious outbreaks, reinforcing the value of early‑life monitoring and internal biosecurity.
Introduction Drives Risk More Than Farm Density
A large cross‑sectional study of Quebec dairy herds found S. Dublin positivity was associated with management practices involving animal purchase and young‑stock raising, while farm density, cow density, and proximity to other dairies were not associated. These results reinforce animal movement as a dominant driver of herd‑level risk in lower‑prevalence settings.
Limited Role of Wild Birds
Field studies in Danish dairy farms found no evidence of Salmonella carriage, including S. Dublin, in common starlings captured directly within infected cattle barns. These findings suggest birds are unlikely to play a significant role in on‑farm transmission, suggesting biosecurity efforts focus on cattle movement and environment.
Barriers to Biosecurity Adoption
Qualitative focus‑group studies in ON and BC among dairy producers identified substantial gaps between awareness of S. Dublin and implementation of biosecurity practices. Low perceived personal risk, cost and labor constraints, unclear responsibilities, and lack of actionable guidance limited adoption of best practices. There was significant uncertainty and variability in how producers interpret and act on a positive test result. Producers with prior outbreak experience showed a shift toward proactive biosecurity, indicating that surveillance must be paired with effective communication and support to drive change.
In Case You Missed It:
- Project report: Investigation of Mycoplasma wenyonii and Candidatus Mycoplasma haemobos in Ontario dairy cattle
- Tips for Testing for Mycoplasma wenyonii and Candidatus Mycoplasma haemobos
- Factsheet: Bovine Theileriosis (Theileria orientalisgenotype Ikeda)
- Factsheet: Asian Longhorned Tick – An Encroaching Threat
NOV 2025 – JAN 2026


